Our planet is surrounded by an incredibly thin envelope of gasses. The part that allows all life to exist is a mere 25,000 feet or a little over 8 kilometres deep – if we go any any higher than this man, and almost all other animals on this planet, suffers from hypoxia and fails to stay alive (without artificial means).
If you imagine the earth the size of an orange with the atmosphere as the ‘skin’ you would have an orange with a skin a mere 3/4 of a millimeter (around 1/30th of an inch) thick.
It is only the fact that the earth is quite large that gives us sufficient air to breathe without choking on all the toxic (to animal life) gasses that form an increasing percentage of it.
Our atmosphere is calculated to have a mass of some 5.148 million million million kilograms (approx 5 thousand million million tons)(1) which sounds quite a lot – and indeed it is a big number.
It is however, ALL there is and has to last both us – all mammals, all insects, reptiles, birds, and plant life as well as microorganisms, for as long as we all live on this planet.
It is important that we do as little damage to it as possible – for many, many reasons.
Very few people ever actually take time to think about what damage we, as members of the human race, could possibly be doing to the very air we need to live. We naturally tend to think that the air has always been there and pretty much always will be and nothing we ever do can change what will be in any significant way.
For the vast percentage of our human history this has pretty much been the case – but no longer. We have already altered and are continuing to alter the composition of the atmosphere – not just on a local scale with the examples of city air pollution and country-wide smogs but on a truly global scale. Anyone remember the Holes in the Ozone Layer?
How can this possibly be?
The answer is as simple as two words: population and lifestyle.

Estimated Human Population for the last 10 000 years
———————————————( Click image to enlarge)————————————–
For ten thousand years mankind lived in insufficient numbers to seriously alter the global environmental balance – and if he looked like starting to, Nature ensured that drought, flood or famine or disease kept the population down to acceptable levels and damage was limited and temporary.
Modern man’s persistence and inventiveness however, has meant that he is now able to overcome most diseases and at the same time produce more and more serious ways to affect larger and larger portions of our environment on ever-increasing levels. Our population simultaneously increased almost exponentially and more and more of us left a subsistence lifestyle, where our local environment gave us what we needed to live, and we started to take what we needed from areas where we were not living. Local economy’s gave way to National, then Empire economies and ultimately to a ‘global’ economy where we took not only from our immediate environment but also from the environment on far-distant continents to enable us to live a somewhat privileged lifestyle. The cumulative effects of this upon our planet’s livable environment began to become more detectable and pervasive – and permanent.
Consider the case of Carbon Dioxide – an essential component gas of our atmosphere.
The gas is toxic to humans at concentrations in excess of 10% while at the same time is essential for growth of all plant life on the planet. Fortunately for us the percentage of this toxic poison has stayed relatively constant for the entire history of human existence at between 200-400 ppmv (parts per million by volume).
Composition of dry atmosphere, by volume.
(ppmv: parts per million by volume)
Gas Volume
Nitrogen (N2) 780,840 ppmv (78.084%)
Oxygen (O2) 209,460 ppmv (20.946%)
Argon (Ar) 9,340 ppmv (0.9340%)
Carbon dioxide (CO2) 390 ppmv (0.039%)
Neon (Ne) 18.18 ppmv (0.001818%)
Helium (He) 5.24 ppmv (0.000524%)
Methane (CH4) 1.79 ppmv (0.000179%)
Krypton (Kr) 1.14 ppmv (0.000114%)
Hydrogen (H2) 0.55 ppmv (0.000055%)
Nitrous oxide (N2O) 0.3 ppmv (0.00003%)
Carbon monoxide (CO) 0.1 ppmv (0.00001%)
Xenon (Xe) 0.09 ppmv (9×10-6%) (0.000009%)
Ozone (O3) 0.0 to 0.07 ppmv (0 to 7×10-6%)
Nitrogen dioxide (NO2) 0.02 ppmv (2×10-6%) (0.000002%)
Iodine (I2) 0.01 ppmv (1×10-6%) (0.000001%)
Ammonia (NH3) trace
Not included in above dry atmosphere:
Water vapor (H2O) ~0.40% over full atmosphere,
typically 1%-4% at surface.
“Each year since global measurements of CO2 began, the amount of carbon dioxide in the atmosphere has increased.
Scientific measurements of levels of CO2 contained in cylinders of ice, called ice cores, indicate that the pre-industrial carbon dioxide level was 278 ppm. That level did not vary more than 7 ppm during the 800 years between 1000 and 1800 A.D.
Atmospheric CO2 levels have increased from about 315 ppm in 1958 to 378 ppm at the end of 2004, which means human activities have increased the concentration of atmospheric CO2 by 100 ppm or 36 percent (since 1800).”(2)
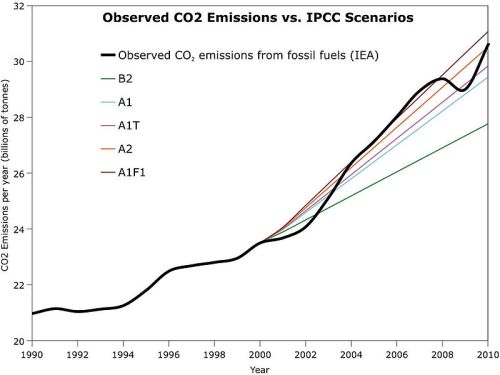
In 2009 it is estimated that human activity pumped almost 30 billion tonnes of CO2 into the air(3). The total amount of CO2 in the air currently is around 390 ppmv or 0.039%. A quick calculation shows that this is roughly: 5148 x 0.00039 x 10^12 ( million million) tonnes, or 2000 billion tonnes. The contribution in 2009 by man was more than 1.4% of the total. There was a minor hiccup after the Global Financial Crisis of 2008 where, for a year or two, we did not actually have a significant increase but we have now resumed the rapidly climbing CO2 production rate.
Humans are therefore now able to increase the amount of atmospheric CO2 by far more than 10% a decade – and the period we do this in still shrinks decade by decade!
The following graph shows this in a somewhat more dramatic fasion:
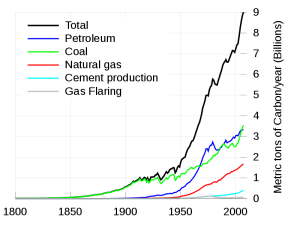
Graph of Carbon (not CO2) Humans put into the atmosphere during the Industrial Revolution to date
Later posts will suggest why this trend MUST be reversed;
.
References:
(1) http://en.wikipedia.org/wiki/Atmosphere_of_Earth
(2) http://www.noaanews.noaa.gov/stories2005/s2412.htm ( NOAA is the US Department of Commerce’s agency responsible for all climate data dissemination to their public – Roughly the Equivalent of Australia’s Bureau of Meteorology).
(3) http://lgmacweb.env.uea.ac.uk/lequere/co2/carbon_budget.htm